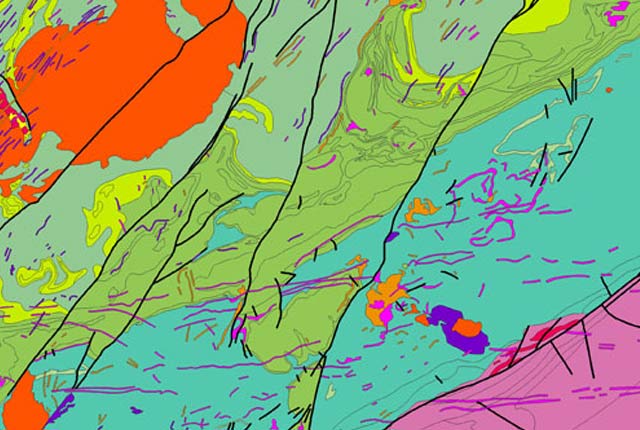
Understanding our Earth
We are a world-leading independent research organisation providing objective, expert geoscientific data, information and knowledge.
Latest news and events

UK Critical Minerals Intelligence Centre – 2026 Showcase
Event on 31/03/2026
Join us for a special live webinar with the team from the Critical Minerals Intelligence Centre showcasing the latest research from 2026 – Copper, the Future of Digital, and What’s next for the Criticality Assessment?

PhD adventures in Copenhagen, Denmark: revealing past recovery processes of tropical forest systems through ancient environmental DNA
12/03/2026
PhD student Chris Bengt visited the University of Copenhagen to carry out very delicate extraction of aeDNA from lake-sediment cores, in the hopes of unlocking the secrets of past volcanic eruptions.
Call for new members and Chair to join the NERC facilities steering committees
25/02/2026
New members are needed to join the committees over the next four years.

Your views wanted – developing a ‘Geothermal energy subsurface data portfolio’
24/02/2026
BGS is aiming to support the growth of the sector by providing the best-available, location-specific geothermal and ground source heat information as an accessible product or service.
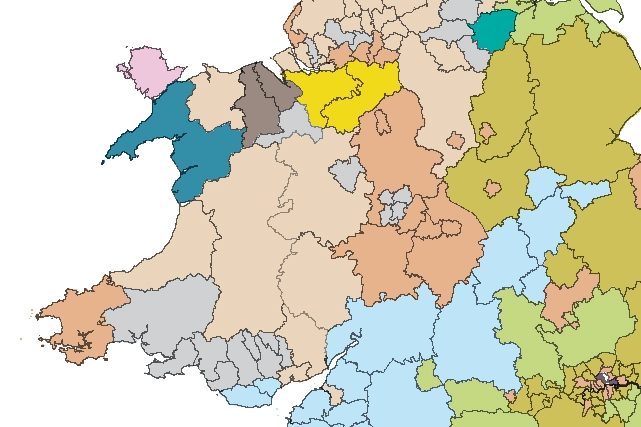
Map of BGS BritPits showing the distribution of worked mineral commodities across the country
18/02/2026
BGS’s data scientists have generated a summary map of the most commonly extracted mineral commodities by local authority area, demonstrating the diverse nature of British mineral resources.

MARC Conference 2025: highlighting the importance of conferences to PhD students
16/02/2026
BGS and University of Nottingham PhD student Paulina Baranowska shares her experience presenting her research on nuclear forensics at her first international conference.

Funding awarded to map the stocks and flows of technology metals in everyday electronic devices
12/02/2026
A new BGS project has been awarded Circular Electricals funding from Material Focus to investigate the use of technology metals in everyday electrical items.

New UK/Chile partnership prioritises sustainable practices around critical raw materials
09/02/2026
BGS and Chile’s Servicio Nacional de Geología y Minería have signed a bilateral scientific partnership to support research into critical raw materials and sustainable practices.

Extensive freshened water confirmed beneath the ocean floor off the coast of New England for the first time
09/02/2026
BGS is part of the international team that has discovered the first detailed evidence of long-suspected, hidden, freshwater aquifers.

Funding secured to help mitigate ground risk in UK construction sector
05/02/2026
The BGS Common Ground project has been awarded new funding to help unlock the value of ground investigation data.

After-school kids’ clubs
Event from 25/02/2026 to 25/03/2026
An after school club for junior geology enthusiasts
Our core challenge areas

Decarbonisation and resource management
Decarbonisation of power production, heat, transport and industry is a major challenge and one that intrinsically involves geoscience.

Multi-hazards and resilience
We work with partners worldwide to enhance understanding of hazards, vulnerability, exposure and risk to ensure our science is useful, usable and used.

International geoscience
BGS is active across the globe delivering institutional strengthening programmes and applied research projects across a wide variety of sectors.

National geoscience
The National Geoscience programme is a forward-looking investment in UK geology that provides relevant, coherent and enriched geoscience knowledge for our stakeholders.

Digital geoscience
The BGS is a data-rich organisation. Our data science and data infrastructure are fundamental to our future research and underpin our strategic challenges.
Map viewers
Data published through map viewers allowing you to reveal more about the ground beneath your feet.

Technologies
Applications, software and online services created by the BGS and our collaborators.

Information hub
Data collections, publications, scanned records and other data gathered by BGS and provided by external organisations.
OpenGeoscience
Accessing maps and downloading data, scans, photos and other information via BGS’s free OpenGeoscience service.

Digital geoscience
The BGS is a data-rich organisation. Our data science and data infrastructure are fundamental to our future research and underpin our strategic challenges.
Digital data licensing and resellers
Providing a wide range of licences for the use and utilisation of our information products by for those interested in an information product and the Open Government Licence is not applicable.

National Geoscience Data Centre
Collecting and preserving geoscientific data, making it available to a wide range of users and communities.

National Geological Repository
Hosting BGS’s collections of borehole cores, cuttings, samples, specimens, and related subsurface information from the UK landmass and continental shelf.

Discovering Geology
Discovering Geology introduces a range of geoscience topics to school-age students and learners of all ages. Explore these pages to discover the fascinating processes and properties that shape our dynamic planet.

Rocks and minerals
Find out more about the differences between rocks and minerals and how they are formed.

Discovering Geology: climate change
What is the difference between weather and climate? what causes the Earth’s climate to change and what are the impacts? Find out more with our Discovery Geology climate change resources.

Earth hazards
The Earth beneath our feet is constantly shifting and moving, and violently with catastrophic and immediate results. Find out more about earth hazards.

Geological processes
Planet Earth is dynamic with a surface that is always changing. Find out about the processes that cause these changes.
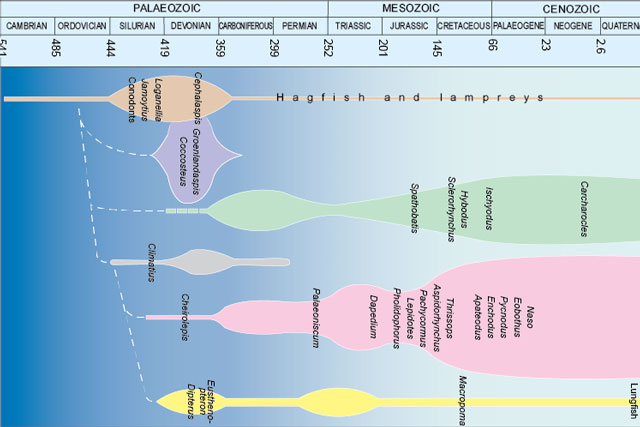
Fossils and geological time
Take a look at the history of the Earth, from its formation over four and a half billion years ago to present times.

About BGS
As the national geological survey, we are the UK’s premier provider of objective and authoritative scientific data, information and knowledge to help society understand our Earth.

Our data and services
The BGS offers a range of services and information for businesses, home-owners, policymakers and the general public.

Working with us
BGS is committed to being an employer of choice and strives to embrace a culture of inclusivity and flexibility.

Offices and locations
Find out more about our offices and facilities located across the UK.

Contact us
Find out more about the BGS and the services we offer through our enquiries and customer services team.